Probes Newsletter 01/2018
Newsletter 01/2018
Make sure you are always up-to-date...
MetaSystems is very proud to announce the XL CBFB/MYH11 plus translocation/dual fusion probe today. Always listening to our customers, we have revitalized this product as a complement to our well proven XL CBFB break apart probe. FISH probes with a break-apart design might easily overlook cryptic insertions of MYH11 into CBFB and vice versa because no separation of flanking regions of CBFB occurs. Translocation/dual fusion FISH probes on the other hand, are indicating this kind of cryptic rearrangement. Furthermore, we are expanding our portfolio with a probe for the detection of rearrangements affecting the TLX3 gene region involved in the development of acute lymphoblastic leukemia.
Please find more detailed information below.
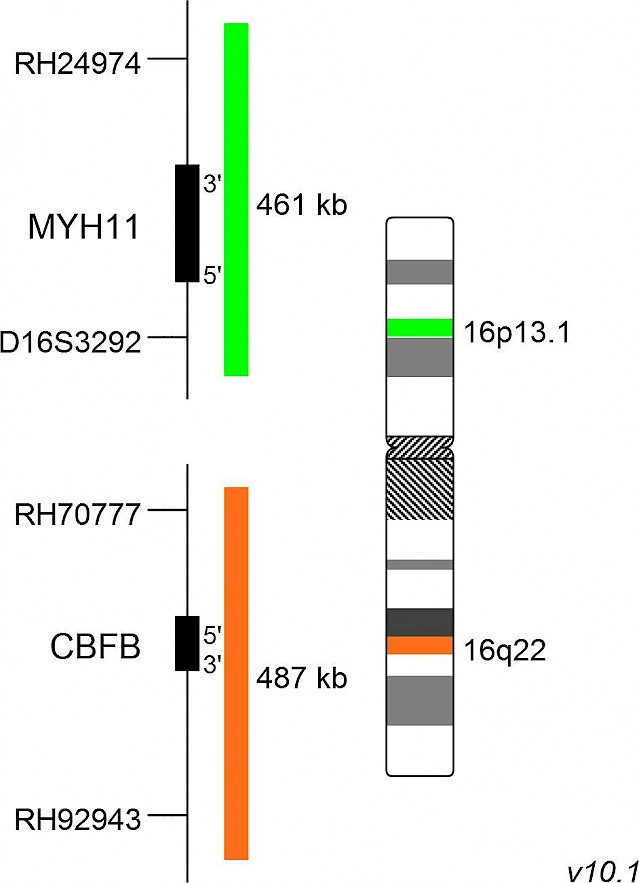
XL CBFB/MYH11 plus
D-5126-100-OG
Clinical Applications: AML
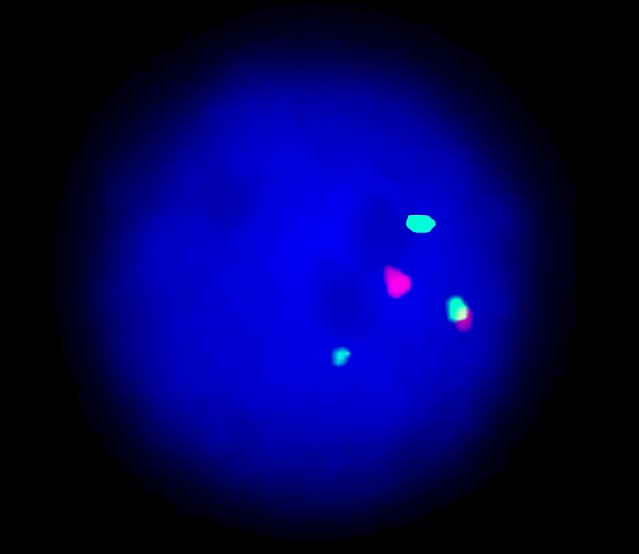
XL CBFB/MYH11 plus is designed as a dual fusion probe. The orange labeled probe spans the breakpoint at 16q22 (CBFB), and the green labeled probe spans the breakpoint at 16p13 (MYH11).
Inv(16)(p13.1;q22) and t(16;16)(p13.1;q22) can be detected in about 10% of young AML patients. Both rearrangements are causing a split of the orange and green signal, resulting in two green-orange colocalization/fusion signals. The remaining normal copies of CBFB and MYH11 are contributing one orange and green signal each. These patients usually have a good prognosis. The rare case of a partial insertion of MYH11 into CBFB is indicated by two green, one orange and one fusion signal as shown above. Insertions into the other direction, CBFB into MYH11, are indicated by two orange, one green and one fusion signal.
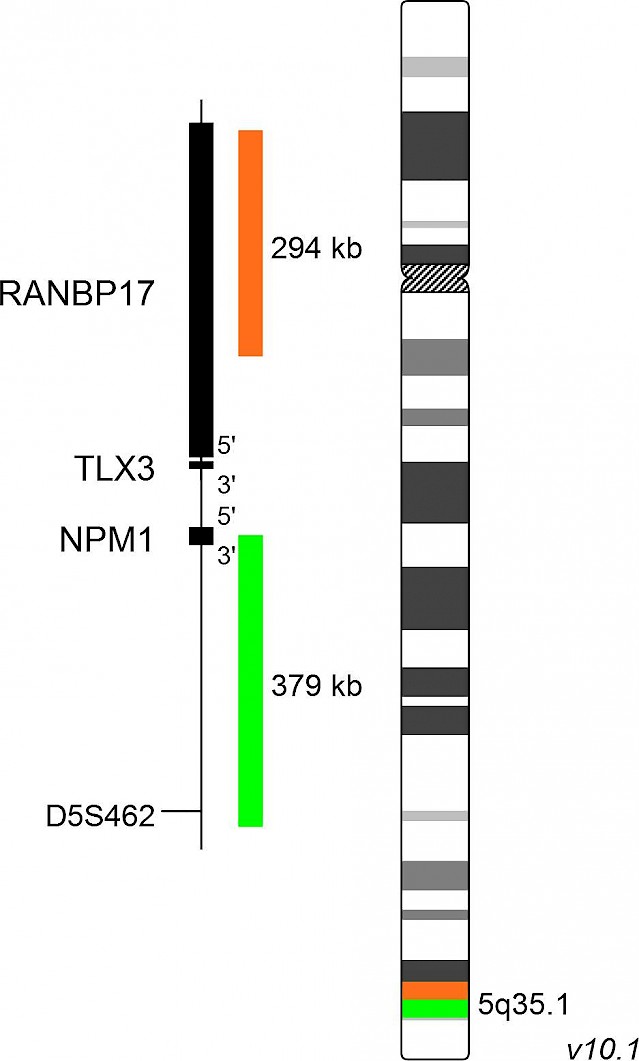
XL TLX3 BA
D-5129-100-OG
Clinical Applications: ALL
XL TLX3 BA is designed as a break apart probe. The orange labeled probe hybridizes proximal to the breakpoint in the TLX3 gene region at 5q35, the green labeled probe hybridizes distal to the breakpoint.
Acute lymphoblastic leukemia (ALL) is the most common childhood cancer type. T-cell acute lymphoblastic leukemia (T-ALL) is an aggressive and quickly progressing type of ALL affecting T-lymphocytes. About 20 % of childhood T-ALL cases are characterized by aberrant expression of TLX3 as a result of t(5;14)(q35;q32). This cryptic translocation juxtaposes TLX3, normally not expressed in T-cells, with the BCL11B gene which is active in T-cells and results in ectopic expression of TLX3. Fluorescence in situ hybridization is a valuable method for the detection of t(5;14)(q35;q32) since cryptic translocations may escape during classical cytogenetic analysis. Furthermore, the broad range of breakpoints in the chromosomal region 14q32 makes the development of efficient PCR-based methods difficult.


