XL ABL1 BA
Break Apart Probe
- Order Number
- D-5148-100-OG
- Package Size
- 100 µl (10 Tests)
- Chromosome
- 099
- Regulatory Status
- IVDD
IVDR Certification
MetaSystems Probes has already certified a wide range of FISH probes, according to IVDR.
This product remains IVDD-certified until further notice.
Discover all IVDR-certified products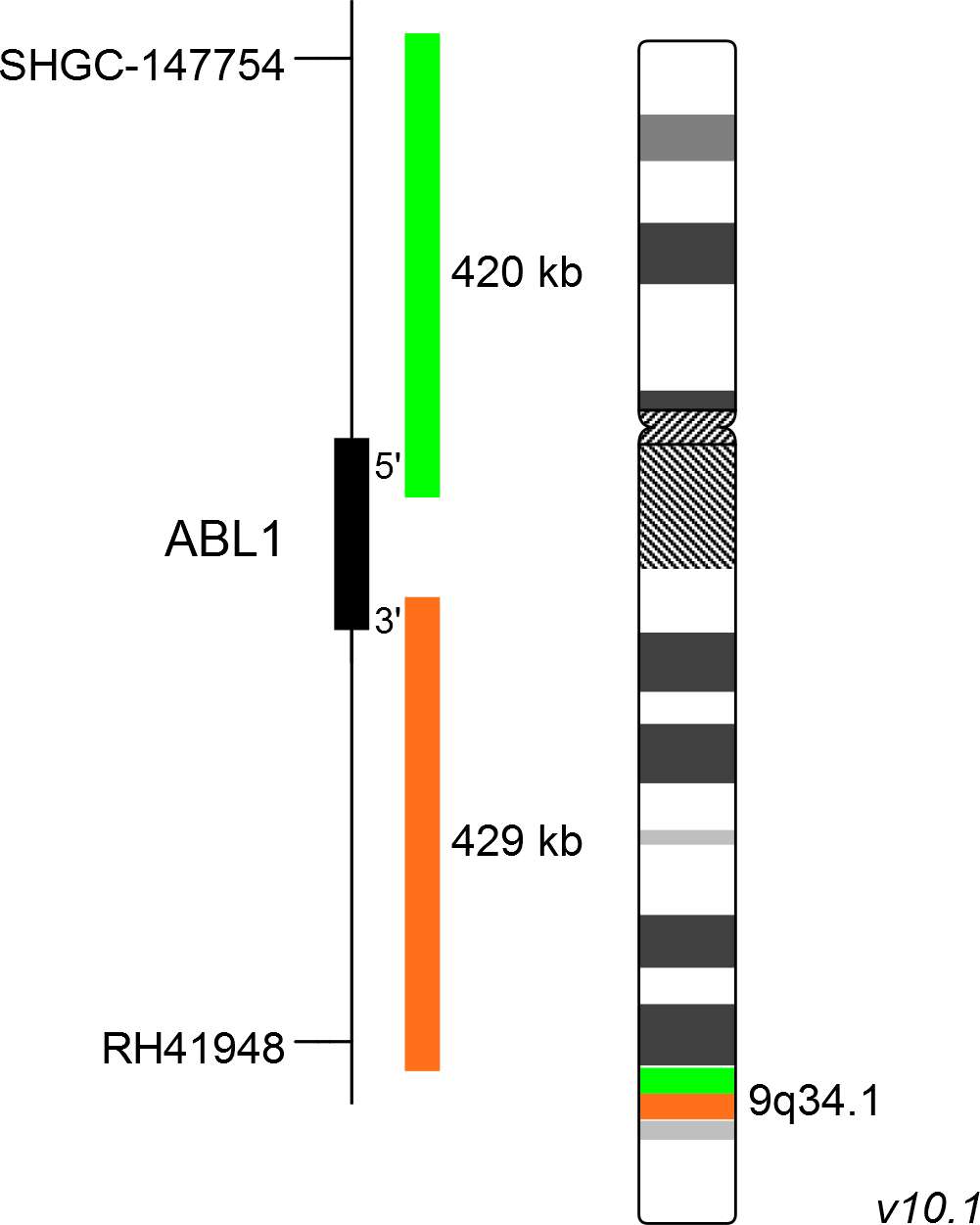
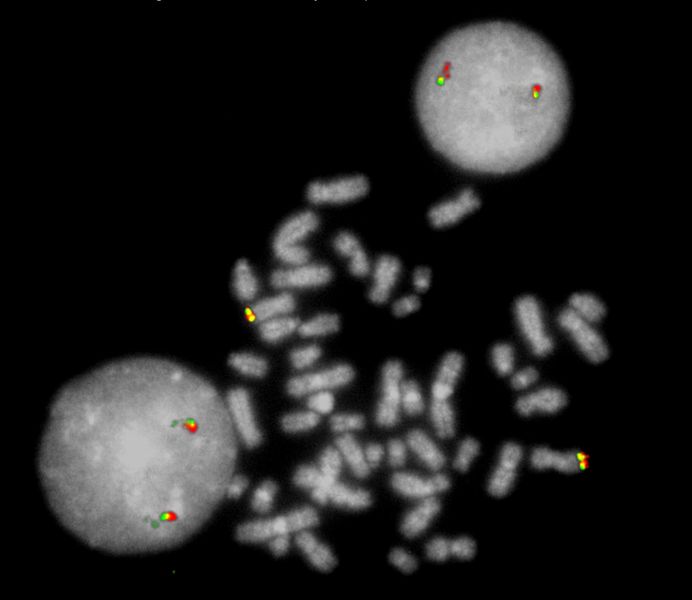
XL ABL1 BA consists of a green-labeled probe hybridizing proximal to the ABL1 gene region at 9q34.1 and an orange-labeled probe hybridizing distal to the ABL1 gene region at 9q34.1.
Probe maps for selected products have been updated. These updates ensure a consistent presentation of all gaps larger than 10 kb including adjustments to markers, genes, and related elements. This update does not affect the device characteristics or product composition. Please refer to the list to find out which products now include updated probe maps.
Probe map details are based on UCSC Genome Browser GRCh37/hg19, with map components not to scale.
Acute lymphoblastic leukemia (ALL) is the most common malignancy in children with a prevalence of approximately 1:1500. Children with Down syndrome have a 10- to 20-fold increased risk of developing acute leukemia (B-cell precursor-ALL, acute myeloid leukemia, and particularly acute megakaryoblastic leukemia).
In 2017, the WHO recognized BCR-ABL1-like ALL, a subtype of B-lymphoblastic leukemia/lymphoma, as a new entity. BCR-ABL1-like ALL, also known as Philadelphia chromosome (Ph)-like ALL, is found in 10-20% of childhood and in 20-30% of all adult B-cell dependent ALL cases. BCR-ABL1-like ALL is characterized by a gene expression profile sharing significant overlap with that of Ph-positive (Ph+) ALL. In contrast to Ph+ ALL, defined by the presence of the BCR-ABL1 fusion resulting from t(9;22)(q34;q11), BCR-ABL1-like cases include a variety of genomic alterations enhancing kinase and cytokine receptor signaling.
Prominent genes involved in the pathogenesis of BCR-ABL1-like ALL are CRLF2, EPOR, JAK2, ABL1, ABL2, CSF1R and PDGFRB. The already known heterogenous 5' fusion partners of ABL1 are CENPC, ETV6, FOXP1, LSM14A, NUP153, NUP214, RANBP2, RCSD1, SFPQ, SNX1, SNX2, SPTAN1 and ZMIZ1. The kinase domain of ABL1 is present in all identified chimeric fusion proteins.
Clinical Applications
- Acute Lymphoblastic Leukemia (ALL)
- Acute Myelogenous Leukemia (AML)
- Chronic Myelogenous Leukemia and Myeloproliferative Neoplasms (CML/MPN)

Normal Cell:
Two green-orange colocalization/fusion signals (2GO).

Aberrant Cell (typical results):
One green-orange colocalization/fusion signal (1GO), one separate green (1G) and orange (1O) signal each resulting from a chromosome break in the relevant locus.
- Tasian et al (2017) Blood 130:2064-2072
- Conant and Czuchlewski (2019) Int J Lab Hematol 41 Suppl 1:126-130
- Jain and Abraham (2019) Arch Pathol Lab Med:doi:10.5858/arpa.2019-0194-RA
Certificate of Analysis (CoA)
or go to CoA Database