XL Smith-Magenis/Miller-Dieker
Deletion Probe
- Order Number
- D-5422-050-OG
- Package Size
- 50 µl (5 Tests)
- Chromosome
- 1717
- Regulatory Status
- IVDD
IVDR Certification
MetaSystems Probes has already certified a wide range of FISH probes, according to IVDR.
This product remains IVDD-certified until further notice.
Discover all IVDR-certified products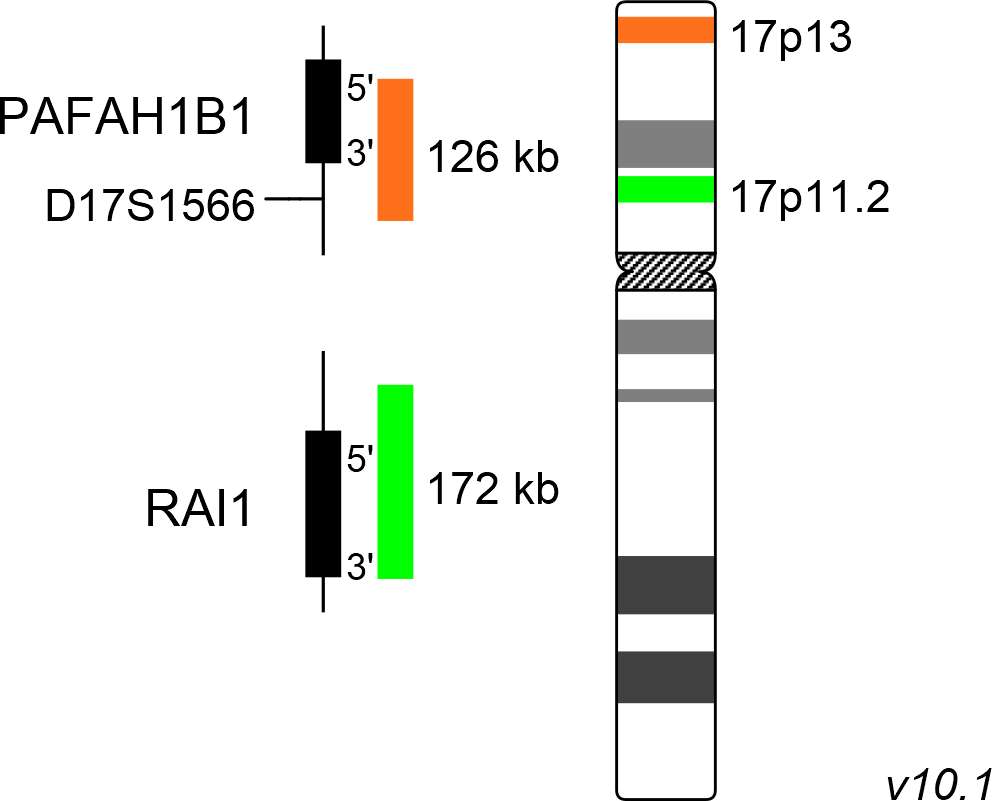
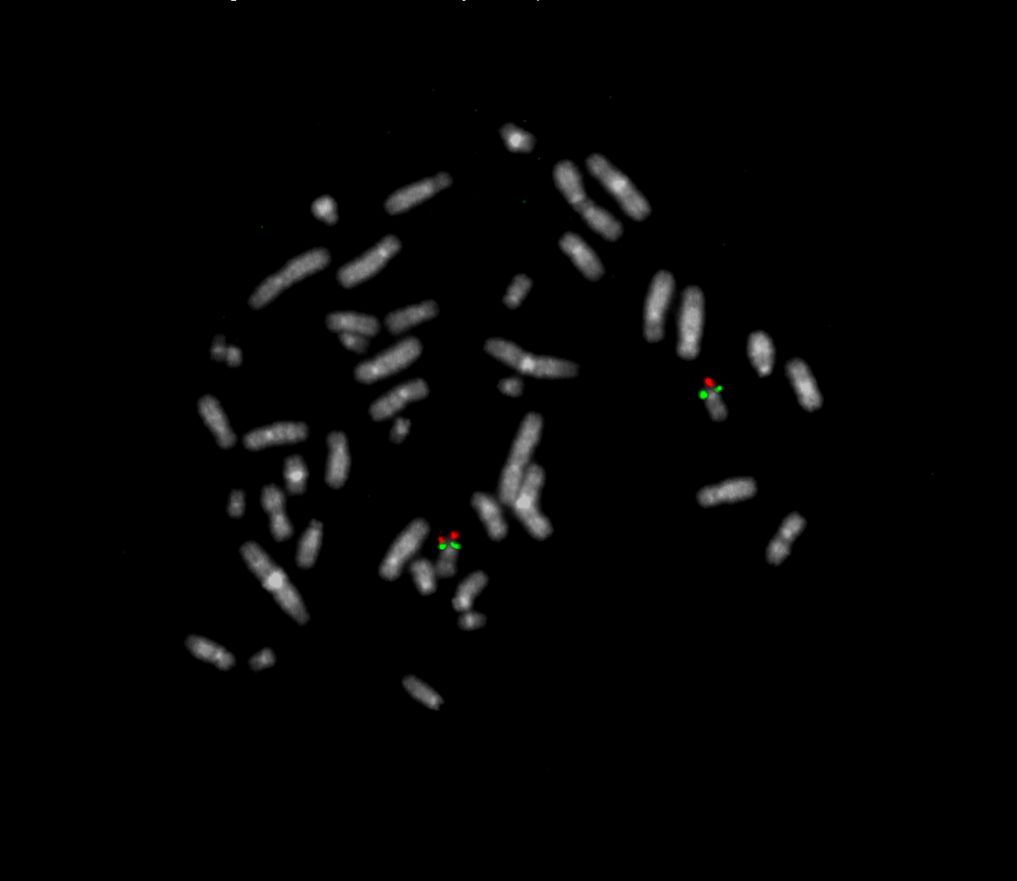
XL Smith-Magenis/Miller-Dieker consists of a green-labeled probe hybridizing to the RAI1 gene region at 17p11.2 and an orange-labeled probe hybridizing to the PAFAH1B1 gene region at 17p13.
Probe maps for selected products have been updated. These updates ensure a consistent presentation of all gaps larger than 10 kb including adjustments to markers, genes, and related elements. This update does not affect the device characteristics or product composition. Please refer to the list to find out which products now include updated probe maps.
Probe map details are based on UCSC Genome Browser GRCh37/hg19, with map components not to scale.
First described in 1982 by Ann C. M. Smith and her colleagues, Smith-Magenis syndrome (SMS) has an estimated prevalence of 1:25.000 newborns. While interstitial deletions in the segment 17p11.2 are the genetic basis of Smith-Magenis syndrome (SMS), deletions of the more distal part 17p13.3 lead to Miller-Dieker syndrome (MDS). SMS is characterized by relatively mild craniofacial and skeletal abnormalities, delay in psycho-motoric development, and specific behavioural disorders. In addition to neuro-behavioural problems, often including self-injurious pattern, SMS patients suffer from a disturbed sleep cycle, as the day and night melatonin secretion cycle is reversed.
MDS patients show a neuro-developmental and structural disorder of the brain, called lissencephaly. The typical walnut structure of the brain containing fissures and gyri is hardly recognizable. Craniofacial dysmorphism, decreased head circumference, delay in psychomotor development and seizures are further symptoms of this syndrome. The SMS inducing deletions of 17p11.2 have no preferential breakpoints, but most patients analyzed show deletions smaller than 4Mb. The key gene responsible for the manifestation of the clinical symptoms of SMS is RAI1 (retinoic acid induced 1), coding for a transcription factor. One of the crucial genes involved in the development of Miller-Dieker lissencephaly and craniofacial dysmorphism is the PAFAH1B1 gene encoding the regulatory subunit of the platelet activating factor acetylhydrolase 1b.
Clinical Applications
- Microdeletion Syndrome (MicroDel)

Normal Signal Pattern:
Two green (2G) and two orange (2O) signals.

Aberrant Signal Pattern:
Two green (2G) and one orange (1O) signal resulting from loss of one orange signal.

Aberrant Signal Pattern:
One green (1G) and two orange (2O) signal resulting from loss of one green signal.
- Elsea (2008) Eur J Hum Genet 16:412-421
- Blazejewski et al (2018) Front Genet 9:8:doi:10.3389/fgene.2018.00080
- Falco et al (2017) Appl Clin Genet 10:85-94
Certificate of Analysis (CoA)
or go to CoA Database